Thermodynamics is known as the study of energy, and how available energy affects reactions and the transformation of matter (Mukherjee 2011). In the context of mineralogy, thermodynamics can tell us which minerals will form under different environmental conditions (Perkins 2022). One example of this in nature is the formation of diamonds and graphite, both of which are polymorphs of pure carbon (East Carbon 2024).
Polymorphism is a property where two minerals have the exact same chemical formula, but different internal crystal structures (Nelson 2019). This also leads to different physical characteristics, such as hardness, solubility, and melting points. As mentioned, diamond and graphite are pure elemental carbon and have a chemical formula of C (Nelson 2019). Despite their identical chemical formulas, they lack any physical resemblance, and diamonds are the hardest known mineral while graphite is one of the softest. This is due to their very different crystal structures (Nelson 2019). Graphite is made up of two dimensional hexagonal sheet structures, where each carbon atom is forming a covalent bond to three other carbon atoms (Wen 2024). These thin layers are held together by weak Van der Waals forces, enabling them to slide very easily past each other. Diamonds on the other hand have a three dimensional tetrahedral crystal structure, where each carbon atom has a strong covalent bond to four other carbons (Figure 1)(Wen 2024).
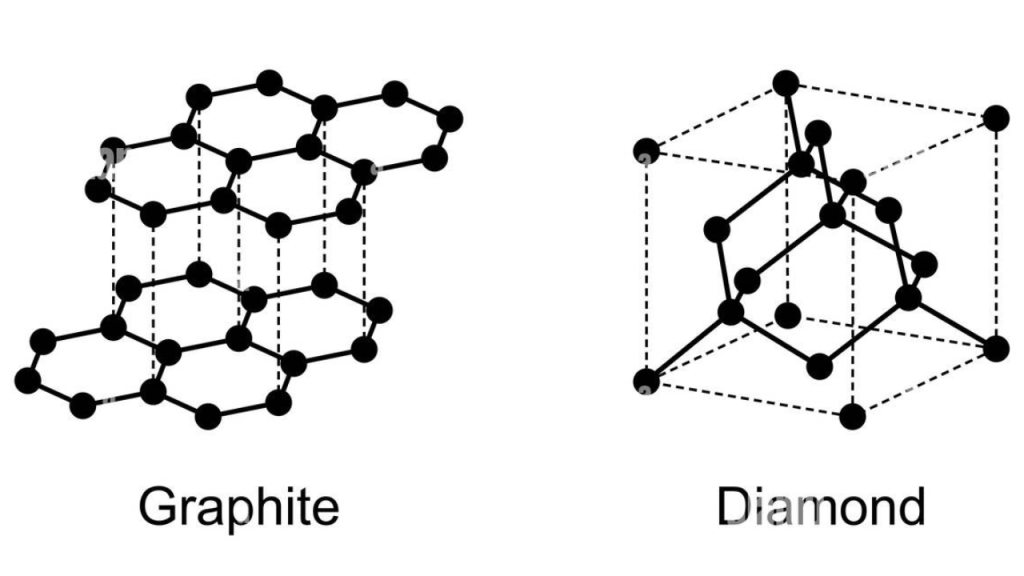
The two main factors that affect which polymorph will form is temperature and pressure (Perkins 2022). Diamonds naturally form 100-300km deep underground in pressures from 5GPa to 120GPa (Hackett 2024). The temperature range is mainly between 1,100°C and 1,500°C (Hackett 2024). In contrast, natural graphite forms higher in the crust at temperatures around 750°C, and pressure ranges from crust levels to 1GPa (Asbury Advanced Materials 2025).
Thermodynamics allows minerologists to understand which minerals will be stable under certain conditions. Typically, minerals are more stable at conditions that are similar to the ones in which they formed, and if they are moved out of those conditions they become thermodynamically unstable (Schulte 2017). This leads to the mineral reacting with the environment and undergoing chemical weathering to become more stable. For example copper oxidizes and turns green when it becomes exposed to oxygen, water and CO2 in the atmosphere (Schulte 2017). So if diamonds are formed so far underground in such harsh conditions, how are they still stable when exposed to atmospheric conditions?
Diamonds are actually at metastable equilibrium under standard conditions (Hackett 2024). This means they are currently in a state where they are energetically stable, but it is not the lowest energy state of the substance. The lowest energy state of pure carbon minerals is graphite, and in thermodynamics all substances want to be at the lowest energy state (White et al. 2020). However, you don’t have to worry about your diamond earring transforming back into graphite anytime soon. Diamonds are unable to convert to graphite once they are mined out of the Earth due to the kinetic energy barrier between diamonds and graphite being too high (Figure 2)(White et al. 2020).
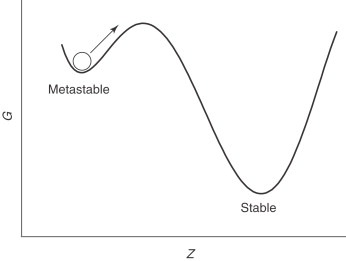
Thermodynamics can also be used to manipulate the carbon polymorphs. In standard conditions with no oxygen present, if you heat up a diamond to temperatures above 1500°C, it can turn into graphite (E. O’Bannon et al. 2020). The heat provides the activation energy needed to overcome the barrier between the metastable and stable equilibrium. The opposite process is also true, where scientists are able to turn graphite into diamonds under extremely high pressure and temperature in labs (D’Haenens-Johansson, Butler, and Katrusha 2022). This mimics the conditions in the Earth’s mantle where diamonds naturally form.
From graphite to diamonds, thermodynamics has given us the ability to explain and manipulate mineral polymorphs. This has introduced many new scientific breakthroughs, such as lab grown diamonds. Who knew that one of the simplest elements, carbon, could create such a variety of minerals.
References
Asbury Advanced Materials. 2025. “Natural Flake Graphite | Asbury Carbons.” Asbury Advanced Materials. 2025. https://www.asbury.com/resources/education/science-of-graphite/natural-flake-graphite/.
D’Haenens-Johansson, Ulrika F. S., James E. Butler, and Andrey N. Katrusha. 2022. “Synthesis of Diamonds and Their Identification.” Reviews in Mineralogy and Geochemistry 88 (1): 689–753. https://doi.org/10.2138/rmg.2022.88.13.
E. O’Bannon, G Xia, F Shi, R Wirth, A King, and L. Dobrzhinetskaya. 2020. “The Transformation of Diamond to Graphite: Experiments Reveal the Presence of an Intermediate Linear Carbon Phase.” Diamond and Related Materials 108 (May): 107876–76. https://doi.org/10.1016/j.diamond.2020.107876.
East Carbon. 2024. “Graphite vs Diamond – a Complete Comparison .” East Carbon. October 14, 2024. https://www.eastcarb.com/graphite-vs-diamond/.
Hackett, William R. 2024. “Diamonds | Research Starters .” EBSCO. 2024. https://www.ebsco.com/research-starters/science/diamonds.
Mukherjee, Swapna. 2011. “Energetics, Thermodynamics and Stability of Minerals.” Applied Mineralogy, 213–30. https://doi.org/10.1007/978-94-007-1162-4_8.
Nelson, Stephen A. 2019. “Twinning, Polymorphism, Polytypism, Pseudomorphism.” Tulane University. January 21, 2019. https://www2.tulane.edu/~sanelson/eens211/twinning.htm.
Perepezko, J.H. 2005. “Phase Transformation.” In Encyclopedia of Advanced Matter Physics, 247–58. Elsevier. https://doi.org/10.1016/B0-12-369401-9/00716-6.
Perkins, Dexter. 2022. “Metamorphic Thermodynamics – Open Petrology.” Open Geology. 2022. https://opengeology.org/petrology/12-thermodynamics-and-metamorphic-equilibria/.
Schulte, Kimberly. 2017. “Weathering.” Geosciences LibreTexts. January 4, 2017. https://geo.libretexts.org/Bookshelves/Geology/Fundamentals_of_Geology_(Schulte)/04%3A_Soil/4.03%3A_Weathering.
Wen, Jianguo. 2024. “Playing with Carbon-Carbon Bonds.” ECS Meeting Abstracts MA2024-02 (11): 1447–47. https://doi.org/10.1149/ma2024-02111447mtgabs.
White, Mary Anne, Samer Kahwaji, Riko Siewert, Joseph A Weatherby, Sergey P Verevkin, Erin R Johnson, and Josef W Zwanziger. 2020. “The Relative Thermodynamic Stability of Diamond and Graphite.” Angewandte Chemie International Edition 60 (3): 1546–49. https://doi.org/10.1002/anie.202009897.
Leave a Reply
You must be logged in to post a comment.