Non-Western medicine and cultural anecdotes have long connected gut and emotional health. Most people have experienced a stomachache when they’re stressed, but what if it is more complicated than that? In the emerging field of gut-brain axis research, evidence suggests that the gut microbiome influences synaptic pathways in the brain and their associated processes.
The gut-brain axis involves bidirectional communication via hormonal, neural, and immune pathways (De Palma et al. 2014). When the body experiences stress, the central nervous system is activated, which alters neuromotor and secretory function in the gut. Disruptions to the gut-brain-axis can alter processes of the hypothalamic-pituitary-adrenal axis (HPA), which regulates stress responses, increasing inflammation and gut permeability (Bertollo et al. 2025). When these changes occur in the gut, endotoxins can leak into the bloodstream and affect the production of neurotransmitters produced in the gut, including serotonin. While most serotonin is produced and used in the gut, serotonin is also vital for mood regulation, and this interaction is of interest in ongoing research. Additionally, overactivation of the HPA causes elevated cortisol levels and subsequently can cause damage in the hippocampus and prefrontal cortex, which are vital for mood regulation and cognitive function.
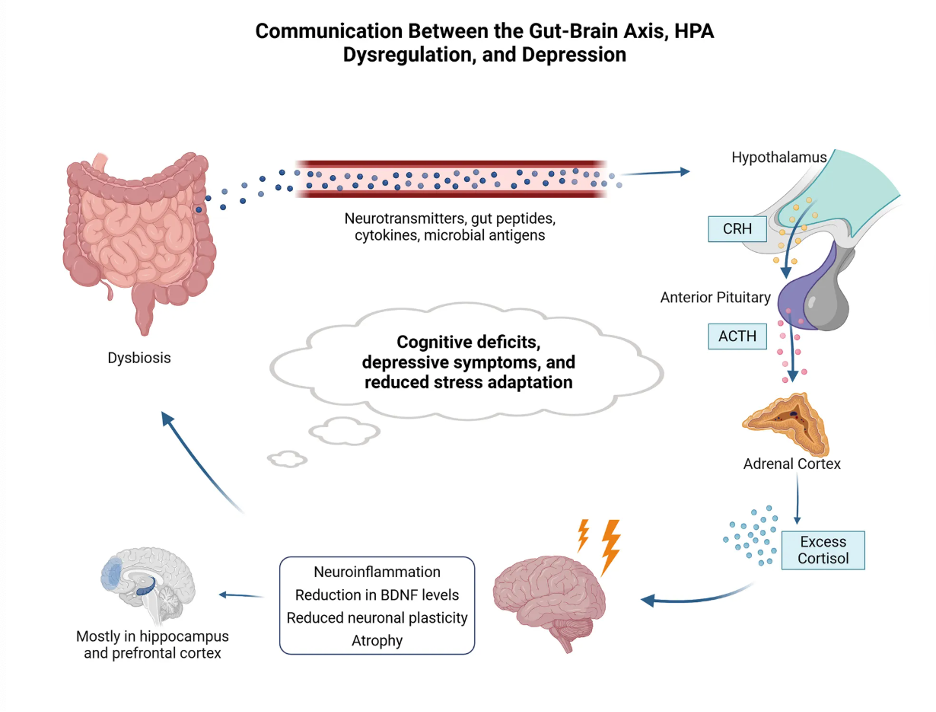
Gastrointestinal diseases have high comorbidities with psychological conditions such as anxiety and depression (Appleton 2018). Additionally, individuals with depression often have increased levels of proinflammatory cytokines, including IL-1β and IL-6. This upregulation of inflammatory cytokines is driven by increased transcription, whereas beneficial metabolites reduce the transcription of proinflammatory cytokines. Major Depressive Disorder treatment using anti-inflammatory drugs has demonstrated success and poses an interesting alternative to traditional treatments.
Antibiotic treatments, which alter gut microbiome composition, have also been shown to impact individuals with a range of psychological disorders and neurodevelopmental conditions. In 2014, Victor and Quigley reported that patients with hepatic encephalopathy – temporarily declined brain function caused by severe liver disease – had symptoms improve with oral antibiotics and laxatives (Victor and Quigley 2014). Antibiotic treatment has also been shown to improve symptoms for individuals with late-onset autism, who are affected by abnormal gut microbiota composition at increased rates (De Palma et al. 2014). There is also anecdotal evidence of antibiotic-induced psychosis, which resolves when treatment ends. These cases present a foundation for the question that many researchers are investigating today: Can we treat the brain by treating the gut? You might have known that stress is causing that gut feeling, but your gut might also be causing that stress.
References
Appleton, Jeremy. 2018. “The Gut-Brain Axis: Influence of Microbiota on Mood and Mental Health.” Integrative Medicine: A Clinician’s Journal 17 (4): 28–32.
Bertollo, Amanda Gollo, Camila Ferreira Santos, Margarete Dulce Bagatini, and Zuleide Maria Ignácio. 2025. “Hypothalamus-Pituitary-Adrenal and Gut-Brain Axes in Biological Interaction Pathway of the Depression.” Frontiers in Neuroscience 19 (February). https://doi.org/10.3389/fnins.2025.1541075.
De Palma, Giada, Stephen M. Collins, Premysl Bercik, and Elena F. Verdu. 2014. “The Microbiota–Gut–Brain Axis in Gastrointestinal Disorders: Stressed Bugs, Stressed Brain or Both?” The Journal of Physiology 592 (14): 2989–97. https://doi.org/10.1113/jphysiol.2014.273995.
Victor, David W., and Eamonn M. M. Quigley. 2014. “Hepatic Encephalopathy Involves Interactions Among the Microbiota, Gut, Brain.” Clinical Gastroenterology and Hepatology 12 (6): 1009–11. https://doi.org/10.1016/j.cgh.2014.01.022.
Leave a Reply
You must be logged in to post a comment.