The integration of quantum physics and biology is new and fascinating. One of the main examples of quantum biology is the effects of quantum tunnelling on DNA mutations. It may not be possible for a person to run through a barrier and come out on the other side. But, in a way, their protons can, and that could end up being more detrimental.
DNA is made up of a phosphate backbone and two pairs of hydrogen bonded nitrogenous base pairs: adenosine with thymine, and guanine with cytosine (Löwdin 1963). When DNA replicates, errors can happen that lead to mutations. These can be caused by external agents like radiation or free radicals (Srivastava 2019). However, there are also spontaneous mutations that arise due to the protons in the hydrogen bonds between the base pairs, and the way this happens is all because of a phenomenon in quantum physics: quantum tunneling (Slocombe et al. 2022). It may seem like an abstract idea because it is not visible at the macroscopic level, but it is a fundamental concept of quantum mechanics (Trixler 2013). Quantum tunnelling occurs when elementary particles, like a proton, permeate an energy barrier without needing to overcome it, as seen in classical physics (Figure 1). One of the main applications of this phenomenon is Scanning Tunneling Microscopy, which allows imaging of resolution at the nanometer scale.
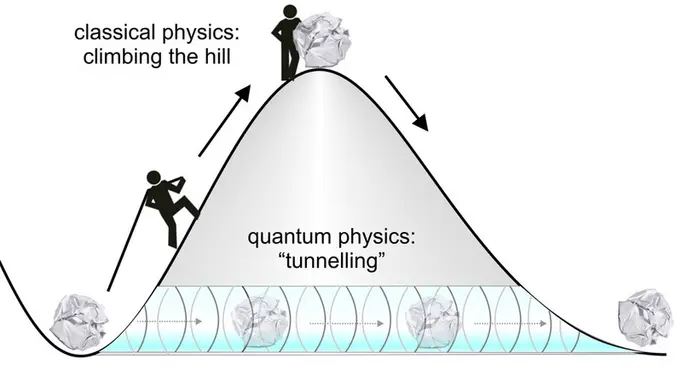
One of the mechanisms behind spontaneous DNA mutations is tautomerism (Srivastava 2019). This is a process that occurs with the protons of the hydrogen bonds binding the nitrogenous bases together (Çelebi et al. 2021). The chemical structure of the base is modified due to a transfer of the proton in the bond, which changes the position of the hydrogen atom, leading to a mutation. How quantum tunnelling allows tautomerization to occur is due to how the potential energy landscape is modelled between the hydrogen bonds. It is a double well potential with a small asymmetry (Figure 2). In classical physics, the protons in the hydrogen bonds are fixed, like a ball at the bottom of the well. In order for the ball to move to another position, it would require enough energy to take it over the top of the hill. This is not the case in quantum mechanics. The protons are not fixed balls, but wavefunctions. They are able to tunnel through these potential energy barriers even if they do not have the minimum amount of energy required to go over the hill as seen in classical physics. This quantum phenomenon is what leads to tautomerization and the mutation of DNA.
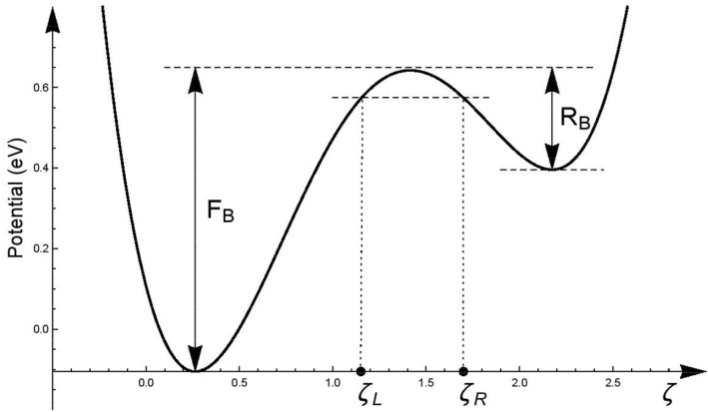
As with many things in the quantum biology world, it can be difficult to determine how much quantum process affect macroscopic systems (Shekaari and Jafari 2020). As computational methods and power have improved over the past few decades, more detailed research has been done to determine whether quantum tunnelling is a significant or trivial aspect of DNA mutation. Work done by Slocombe et al. (2022) shows that the contribution of quantum tunnelling in the proton transfer rate between guanine and cytosine is several orders of magnitude larger than the classical physics model of overcoming the energy barrier. Although the significance of quantum tunnelling may be debated in DNA mutations, what is undeniable is how wondrous the intersection between quantum physics and biology can be.
References
Çelebi, Gizem, Elif Özçelik, Emre Vardar, and Durmuş Demir. 2021. “Time Delay during the Proton Tunneling in the Base Pairs of the DNA Double Helix.” Progress in Biophysics and Molecular Biology 167 (December): 96–103. https://doi.org/10.1016/j.pbiomolbio.2021.06.001.
“Electrons Caught in the Act of Tunnelling.” n.d. Accessed February 22, 2026. https://www.mpg.de/549767/electrons-caught-in-the-act-of-tunnelling.
Löwdin, Per-Olov. 1963. “Proton Tunneling in DNA and Its Biological Implications.” Reviews of Modern Physics 35 (3): 724–32. https://doi.org/10.1103/RevModPhys.35.724.
Shekaari, Ashkan, and Mahmoud Jafari. 2020. “Modeling the Action of Environment on Proton Tunneling in the Adenine–Thymine Base Pair.” Progress in Biophysics and Molecular Biology 150 (January): 98–103. https://doi.org/10.1016/j.pbiomolbio.2019.07.002.
Slocombe, Louie, Marco Sacchi, and Jim Al-Khalili. 2022. “An Open Quantum Systems Approach to Proton Tunnelling in DNA.” Communications Physics 5 (1): 109. https://doi.org/10.1038/s42005-022-00881-8.
Srivastava, Ruby. 2019. “The Role of Proton Transfer on Mutations.” Frontiers in Chemistry 7 (August): 536. https://doi.org/10.3389/fchem.2019.00536.
Trixler, Frank. 2013. “Quantum Tunnelling to the Origin and Evolution of Life.” Current Organic Chemistry 17 (16): 1758–70. https://doi.org/10.2174/13852728113179990083.
Leave a Reply
You must be logged in to post a comment.