There is a type of bacteria that lives quietly on your skin… and sometimes, it fights back. Staphylococcus aureus, commonly known as staph, is a Gram-positive, spherical bacterium belonging to the staphylococcaceae family (Taylor and Unakal 2023). It is frequently found on the skin and in the upper respiratory tract of healthy individuals. In many cases, its presence is considered a carrier state, which means it exists as part of the body’s normal flora without causing harm (Chmielowiec-Korzeniowska et al. 2020). However, when the skin barrier is broken through cuts, incisions, abrasions, or surgical wounds, this otherwise harmless bacteria can enter the body and cause an infection. When the skin barrier is compromised, Staphylococcus aureus can enter underlying tissues and begin to multiply. Once inside the body, the bacteria can trigger localized infections that often present as red, swollen, and painful areas of the skin. These infections often develop into boils, abscesses, or even cellulitis, and are sometimes accompanied by pus formation and fever in more severe, untreated cases (Taylor and Unakal 2023). Under normal circumstances, staph infections can be effectively treated with commonly prescribed antibiotics. Most mild to moderate staphylococcal skin infections resolve without complications (Taylor and Unakal 2023).
However, not all staph infections respond to standard antibiotic therapy. Some strains have developed resistance to methicillin and other widely used antibiotics. These strains are known as Methicillin-Resistant Staphylococcus aureus, or MRSA, and has proven to be an emerging challenge in both the healthcare and pharmaceutical industry. Primarily spreading through direct skin-to-skin contact or contact with contaminated surfaces or shared items, MRSA can also affect individuals asymptomatically, serving as vectors for transmission (Taylor and Unakal 2023). Additionally, MRSA can create biofilms, which are structures of bacterial communities shielded in an extracellular matrix, that enhances its persistence on tissues (Shree et al. 2023). The treatment of MRSA is dependent on the severity of the infection, mild skin infections may only require drainage and wound care, while more invasive cases need antibiotics that are effective against resistant strains, such as vancomycin or doxycycline (Siddiqui and Koirala 2023).
Antibiotic resistance in Staphylococcus aureus emerges through genetic adaptation and evolutionary pressure (Fait, Andersson, and Ingmer 2023). b-lactam antibiotics such as methicillin typically target penicillin-binding proteins (PBPs), these are transpeptidase enzymes involved in bacterial cell wall synthesis. By binding to these proteins, the antibiotics disrupt the cross-linking of the peptidoglycan layers in the cell wall, weakening the structure and leading to cellular death (Bush and Bradford 2016). However, MRSA strains possess the mecA resistance gene (Figure 1), which encodes a different penicillin-binding protein known as PBP2a.
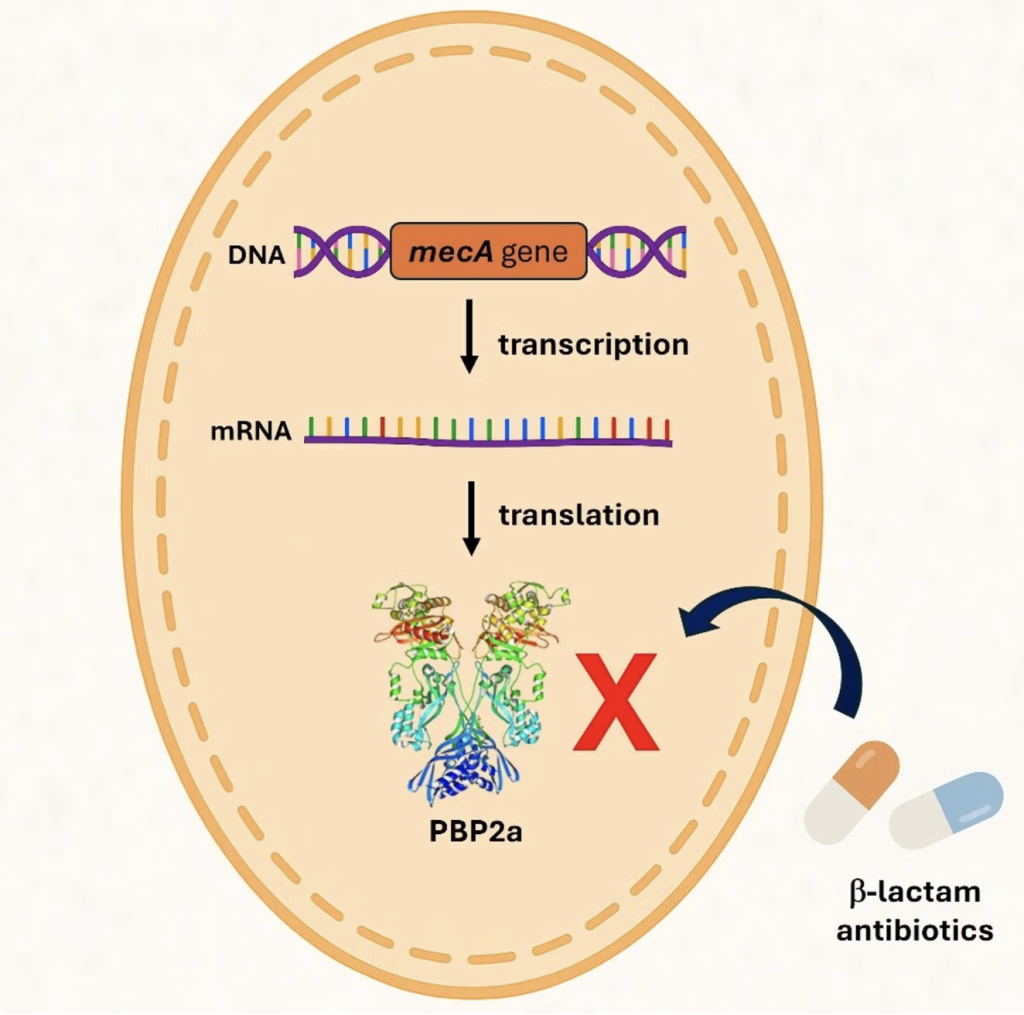
Figure 1. Genetic Mechanism of the mecA gene in MRSA. The gene encodes PBP2a, as opposed to the original PBPs targeted by b-lactam antibiotics, PBP2a has a lower affinity these antimicrobials. b-lactam antibiotics inhibit bacterial formation by binding to PBPs and disrupting cell wall synthesis. Due to the presence of PBP2a, Staphylococcus aureus is able to bypass the inhibition, allowing cell wall synthesis in the presence of b-lactam antibiotics (“Antimicrobial Resistance and the MRSA Crisis: A Silent Pandemic Demanding Urgent Global Action” 2025).
This protein has a decreased affinity for b-lactam antibiotics, which allows MRSA to continue synthesizing its cell wall even in the presence of antibiotics. The mecA gene is located on a mobile genetic element known as the staphylococcal cassette chromosome mec, which enables horizontal gene transfer between bacterial populations. With frequent antibiotic use, selective pressure favors the survival and reproduction or resistant strains over susceptible ones (Fergestad et al. 2020).
In conclusion, MRSA shows how Staphylococcus aureus can adapt and grow under selective pressure to become resistant to antibiotics due to widespread use. Resistance to antimicrobials poses a significant threat to public health, increasing infection severity, making treatment methods more complex, and increasing the risk of transmission within communities and hospitals.
References
“Antimicrobial Resistance and the MRSA Crisis: A Silent Pandemic Demanding Urgent Global Action.” 2025. Selectscience.net. 2025. https://www.selectscience.net/article/antimicrobial-resistance-and-the-mrsa-crisis-a-silent-pandemic-demanding-urgent-global-action.
Bush, Karen, and Patricia A. Bradford. 2016. “β-Lactams and β-Lactamase Inhibitors: An Overview.” Cold Spring Harbor Perspectives in Medicine 6 (8): a025247. https://doi.org/10.1101/cshperspect.a025247.
Chmielowiec-Korzeniowska, Anna, Leszek Tymczyna, Łukasz Wlazło, Bożena Nowakowicz-Dębek, and Beata Trawińska. 2020. “Staphylococcus Aureus Carriage State in Healthy Adult Population and Phenotypic and Genotypic Properties of Isolated Strains.” Advances in Dermatology and Allergology 37 (2): 184–89. https://doi.org/10.5114/ada.2020.94837.
Fait, Anaëlle, Dan I. Andersson, and Hanne Ingmer. 2023. “Evolutionary History of Staphylococcus Aureus Influences Antibiotic Resistance Evolution.” Current Biology: CB 33 (16): 3389-3397.e5. https://doi.org/10.1016/j.cub.2023.06.082.
Fergestad, Marte Ekeland, Gro Anita Stamsås, Danae Morales Angeles, Zhian Salehian, Yngvild Wasteson, and Morten Kjos. 2020. “Penicillin‐Binding Protein PBP2a Provides Variable Levels of Protection toward Different β‐Lactams in Staphylococcus Aureus RN4220.” MicrobiologyOpen 9 (8). https://doi.org/10.1002/mbo3.1057.
Shree, Pallee, Chandra Kant Singh, Kushneet Kaur Sodhi, Jaya Niranjane Surya, and Dileep Kumar Singh. 2023. “Biofilms: Understanding the Structure and Contribution towards Bacterial Resistance in Antibiotics.” Medicine in Microecology 16 (100084): 100084. https://doi.org/10.1016/j.medmic.2023.100084.
Siddiqui, Abdul, and Janak Koirala. 2023. “Methicillin Resistant Staphylococcus Aureus (MRSA).” National Library of Medicine. StatPearls Publishing. April 2, 2023. https://www.ncbi.nlm.nih.gov/books/NBK482221/.
Taylor, Tracey A, and Chandrashekhar G Unakal. 2023. “Staphylococcus Aureus Infection.” National Library of Medicine. StatPearls Publishing. 2023. https://www.ncbi.nlm.nih.gov/books/NBK441868/.
Leave a Reply
You must be logged in to post a comment.