Subjective time perception refers to the cognitive and neural processes that allow individuals to estimate and experience the passage of time (Fontes et al. 2016). This phenomenon is framed within the pacemaker–accumulator model, known as the internal clock theory proposed by Gibbons et al. (1984). The theory proposes that a pacemaker generates pulses that pass through an attention-controlled gate and are accumulated to represent the perceived duration of an event. These accumulated pulses are then temporarily stored in working memory and compared to previously stored durations in reference memory, allowing individuals to judge how much time has passed. Importantly, the rate of this pacemaker is not fixed, as it is modulated by factors such as arousal, attention, and most prominently, circadian rhythm, which is the body’s endogenous 24‑hour cycle (Lake et al. 2016; Figure 1). When the internal pacemaker speeds up, more pulses accumulate and time feels elongated; whereas when it slows, fewer pulses are counted and time feels compressed. As circadian rhythm helps regulate pacemaker speed, any disruption to this rhythm can distort subjective time.
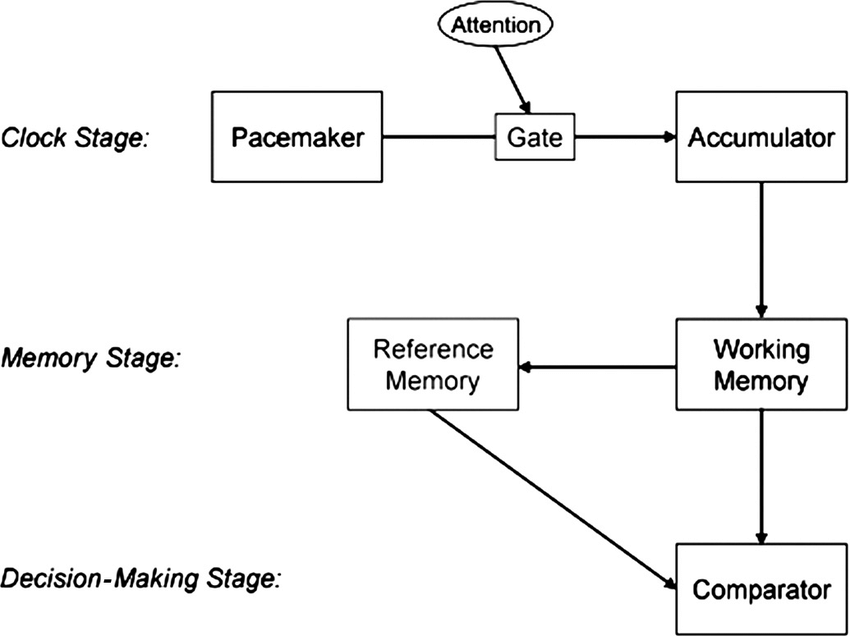
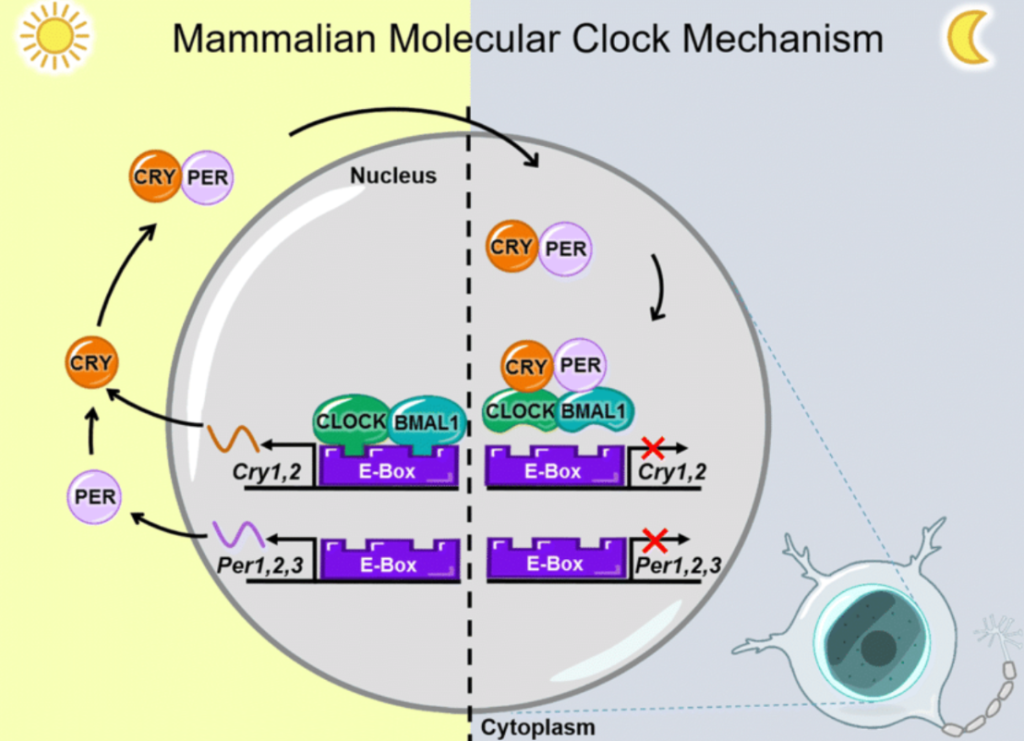
Interestingly, daylight saving time (DST) influences time perception through misaligning with one’s natural circadian rhythm. Although widely adopted by many countries, this practice misaligns social time with biological time, creating a temporary but meaningful shift in circadian rhythm (Romigi et al. 2025). At the biological level, circadian disruption from DST originates from the transcription–translation feedback loop (TTFL) that is responsible for generating the 24 hour rhythms in the body and is coordinated by the suprachiasmatic nucleus (SCN), which is a small cluster of neurons in the hypothalamus known as the body’s master clock that receives direct input from the eyes. In this loop, circadian locomotor output cycles kaput (CLOCK) and BMAL1 are transcription factors that activate the Period (PER) and Cryptochrome (CRY) genes in nearly every cell in the human body. As PER and CRY proteins accumulate, they feed back to inhibit CLOCK–BMAL1, pausing their own production until they naturally degrade and the cycle restarts. This molecular rhythm sets the timing of sleep, hormone release, body temperature, and cognitive performance. As the SCN is synchronized by light, morning sunlight normally shifts the TTFL earlier and evening light shifts it later. When clocks jump forward for DST, social time changes instantly but environmental light does not, leaving the SCN and TTFL running on the previous day’s schedule. This creates a temporary state of internal desynchronization, where biological time lags behind clock time, disrupting sleep, attention, and the cognitive processes that shape subjective time perception. (Fan et al. 2021; Figure 2).
To showcase such effects, Barnes et al. (2009) analyzed 23 years of U.S. mining injury records and found that on the Monday following the spring DST shift, workplace injuries increased by 5.7% and injury severity rose by 67.6%. These findings demonstrate that even a one‑hour circadian phase from DST can produce inconsistent perceptions of time, leading to harmful consequences. Thus, DST is not just an inconvenience for certain people, but also a biologically consequential perturbation for the human body. By altering circadian timing, it distorts subjective time perception, in turn impairing cognitive control, and producing measurable health and safety risks.
Work Cited
Barnes, Christopher M., and David T. Wagner. 2009. “Changing to Daylight Saving Time Cuts into Sleep and Increases Workplace Injuries.” Journal of Applied Psychology (US) 94 (5): 1305–17. https://doi.org/10.1037/a0015320.
Fan, Xu-Li, Ying Song, Dong-Xu Qin, and Pei-Yao Lin. 2023a. “Regulatory Effects of Clock and Bmal1 on Circadian Rhythmic TLR Expression.” International Reviews of Immunology 42 (2): 101–12. https://doi.org/10.1080/08830185.2021.1931170.
Fan, Xu-Li, Ying Song, Dong-Xu Qin, and Pei-Yao Lin. 2023b. “Regulatory Effects of Clock and Bmal1 on Circadian Rhythmic TLR Expression.” International Reviews of Immunology 42 (2): 101–12. https://doi.org/10.1080/08830185.2021.1931170.
Fontes, Rhailana, Jéssica Ribeiro, Daya S. Gupta, et al. 2016. “Time Perception Mechanisms at Central Nervous System.” Neurology International 8 (1): 5939. https://doi.org/10.4081/ni.2016.5939.
Lake, Jessica I., Kevin S. LaBar, and Warren H. Meck. 2016. “Emotional Modulation of Interval Timing and Time Perception.” Neuroscience & Biobehavioral Reviews 64 (May): 403–20. https://doi.org/10.1016/j.neubiorev.2016.03.003.
Rhoads, Megan K., Vikhram Balagee, and S. Justin Thomas. 2020. “Circadian Regulation of Blood Pressure: Of Mice and Men.” Current Hypertension Reports 22 (6): 40. https://doi.org/10.1007/s11906-020-01043-3.
Romigi, Andrea, Valentina Franco, Egeria Scoditti, et al. 2025. “The Effects of Daylight Saving Time and Clock Time Transitions on Sleep and Sleepiness: A Systematic Review.” Sleep Medicine Reviews 84 (December): 102161. https://doi.org/10.1016/j.smrv.2025.102161.
Wencil, Elaine B., H. Branch Coslett, Geoffrey Aguirre, and Anjan Chatterjee. “Carving the Clock at Its Component Joints: Neural Bases for Interval Timing | Journal of Neurophysiology | American Physiological Society.” 2009. Accessed March 2, 2026. https://journals.physiology.org/doi/full/10.1152/jn.00029.2009.
Leave a Reply
You must be logged in to post a comment.